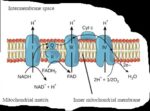
The Electron Transport System (ETS) or Electron Transport Chain (ETC) is a series of protein complexes and electron carriers found in the inner membrane of mitochondria in eukaryotic cells (and in the plasma membrane of prokaryotes). It plays a crucial role in cellular respiration, particularly in the production of ATP (adenosine triphosphate), the energy currency of the cell.
Key Functions of the ETS/ETC:
1. Production of ATP: The main function of the ETC is to produce ATP through a process called oxidative phosphorylation.
2. Transfer of Electrons: Electrons are transferred through a series of complexes (I to IV), moving from higher to lower energy states, releasing energy at each step.
3. Generation of Proton Gradient: The energy released from electron transfer is used to pump protons (H⁺) across the inner mitochondrial membrane, creating a proton gradient (also called the proton motive force).
4. Reduction of Oxygen: At the end of the ETC, electrons combine with protons and oxygen to form water, making oxygen the final electron acceptor.
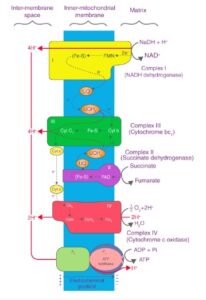
Components of the Electron Transport Chain:
The ETC consists of four protein complexes (Complex I-IV) and mobile electron carriers (ubiquinone, also known as coenzyme Q, and cytochrome c).
1. Complex I (NADH Dehydrogenase): Accepts electrons from NADH, transferring them to ubiquinone. It also pumps protons into the intermembrane space.
2. Complex II (Succinate Dehydrogenase): Accepts electrons from FADH₂ (produced in the citric acid cycle), and transfers them to ubiquinone, but unlike Complex I, it does not pump protons.
3. Ubiquinone (Coenzyme Q): A mobile carrier that transports electrons from Complexes I and II to Complex III.
4. Complex III (Cytochrome bc₁ complex): Transfers electrons from ubiquinone to cytochrome c and pumps protons across the membrane.
5. Cytochrome c: Another mobile carrier that transfers electrons between Complex III and Complex IV.
6. Complex IV (Cytochrome c oxidase): Transfers electrons to oxygen, reducing it to water, while pumping additional protons into the intermembrane space.
Role of the Proton Gradient:
The proton gradient generated by the ETC creates a high concentration of protons in the intermembrane space compared to the mitochondrial matrix. This gradient drives the synthesis of ATP through **ATP synthase**, an enzyme that uses the energy of the protons flowing back into the matrix to convert ADP into ATP. This process is called **chemiosmosis**.
Overview of the Process:
1. Electrons from NADH and FADH₂ (generated in the Krebs cycle) are transferred through the complexes in the ETC.
2. As electrons move through the chain, protons are pumped across the inner mitochondrial membrane.
3. The proton gradient powers ATP synthase, producing ATP.
4. Oxygen serves as the final electron acceptor, forming water.
Importance of the Electron Transport Chain:
– The ETC is the most significant source of ATP in aerobic organisms, generating about 34 ATP molecules per glucose molecule in the overall process of cellular respiration.
– If oxygen is unavailable (anaerobic conditions), the ETC cannot function, which forces cells to rely on less efficient methods like fermentation for energy production.
In summary what we can say like the Electron Transport Chain is a critical part of the process by which cells convert the energy stored in nutrients into usable chemical energy (ATP). Its efficient function is essential for energy production in both eukaryotic and prokaryotic organisms.